Please use the form below to submit comments. Also provide an e-mail address and name. Your e-mail address and/or name will be used only to communicate with you about this or future comments you may submit. I am particularly keen to receive references to published material that contradicts the assertions and arguments I have made.
By submitting the above comment, I grant to Ross Alan Hangartner the right to incorporate the comment in full or in part, literally, paraphrased, or conceptually, as he sees fit, into State of Pain or other writings that he may create in the future. However, I don't grant permission to include my name or e-mail address, or to use them in any other way than to contact me for follow-up. I understand that by submitting the comment I acquire no right of any kind in State of Pain or other writings of Ross Alan Hangartner.
Last updated: Sat, Feb 22, 2025

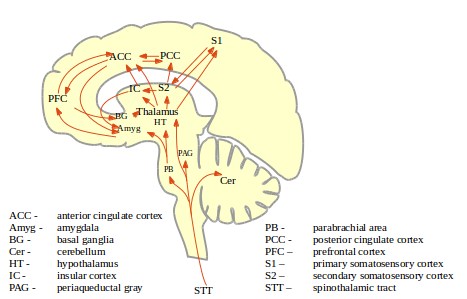
The parabrachial nucleus or PB is found in the pons of the brain stem. (See details about the PB.) It receives lamina I input and a small amount of lamina V input from the dorsal horn. It feeds the thalamus, hypothalamus, and the amygdala.
The periaqueductal gray or PAG is located in the midbrain of the brain stem. (See details about the PAG.) It receives input from lamina I and a small amount of lamina VII-VIII input from the dorsal horn. It feeds the thalamus and hypothalamus.
The cerebellum is commonly active in pain studies. It is not known what its role is in pain perception, but it is believed that it may be more concerned with descending modulation of pain intensity than with pain perception itself.1
The thalamus is a central structure in the brain. It sits atop the midbrain, that is, the top end of the brain stem, and connects extensively to all areas of the cortex as well as to limbic structures and the brain stem. It contains a hub for each of the senses except smell, and it is extensively connected to the hippocampus, which is responsible for creating memories. The thalamus is important in regulating sleep and wakefulness.
Lesion studies have indicated that loss of the somatosensory cortex has little effect on the affective (unpleasantness) perception of pain. This supports the idea that the thalamus and regions in the brain stem and limbic areas of the brain dominate the affective perception of pain. These "lower" areas of the brain are evolutionarily much older than the cortex, which suggests that the sense of pain was a critical sense in the early development of vertebrates.2
The hippocampus is usually regarded as a part of the limbic system, and is sometimes included as part of the basal ganglia. It is heavily involved in transferring information from temporary short-term memory (which occurs in the prefrontal cortex) into long-term memory. In other words, it creates episodic memories. It transmits some signal or signals that seem to make the mind rehearse important information until permanent storage takes place.3
The primary somatosensory cortex (S1 on the diagram, Figure 1: Ascending paths of the pain matrix) and the secondary somatosensory cortex (S2 on the diagram, Figure 1: Ascending paths of the pain matrix) are two areas of the cortex involved in identifying and localizing areas of the body. Each is organized somatotopically, meaning that physical areas of the somatosensory cortex map to physical areas of the body. They are activated by innocuous as well as by noxious input.
You may be familiar with an image of the brain that shows a distorted human body along a slice through the brain. (The slice is "coronal," or across the width of the brain.) The image is called a "homunculus," and shows how the somatosensory cortex and the body correspond. The homunculus is distorted because different areas of the body are innervated more and less densely. For example, the hand is more densely innervated than the arm. (See Pain Processing in the Central Nervous System for more about the somatosensory cortex.)
It is likely that they are both involved in determining the location, duration, and intensity of painful stimuli. This view is reinforced by the report of a patient whose S1 and S2 areas were damaged by a stroke. This patient reported that they felt an unpleasant sensation when subjected to a painful stimulus, but couldn't identify its location or describe the nature of the stimulus. (This kind of evidence is called lesion evidence, the lesion in this case being the stroke damage.) This observation also suggests that the unpleasantness (sometimes called affect) of pain must come from other brain regions.
The cingulate cortex lies on top of the corpus callosum, the large tract of axons that connect the left and right hemispheres of the brain. Just below the corpus callosum are the thalamus, midbrain, and basal ganglia. Thus the cingulate cortex sits between the visible neocortex and the older parts of the brain. It is part of the cortex, and is usually considered part of the limbic system.
The anterior cingulate cortex (ACC) is the more anterior half of the cingulate cortex. It abuts the frontal cortex. Nociceptive neurons have been found in the human ACC, and painful stimulation of a human subject induces potentials (detectable by EEG).
The ACC is believed responsible for assessing the unpleasantness (also called the affect) of painful stimuli. Lesion evidence for this view comes from the results of cingulotomies, that is, cuts through the cingulate cortex, which are sometimes performed for otherwise-intractable pain. Patients who have had this procedure are still aware of pain, but no longer find it to be unpleasant.
Additional evidence of this role for the ACC comes from imaging studies of patients who respond to hypnotic suggestion that reduces their perception of painfulness. (Hypnotic Analgesia.) In these patients, the ACC is less active when they perceive less pain. Another study showed that when subjects reported less-unpleasant pain, fewer opioid receptors in the ACC were activated, a sign of less neural activity in the ACC.
A 2001 study showed that the activition of mu-opioid receptors within the ACC correlated well with the "affective" component of the McGill Pain Questionnaire. (See The McGill Pain Questionnaire.) Pain learning experiments with rats show that when the ACC is inactivated, rats do not learn to avoid painful situations. This suggests that, at any rate in rats, the ACC is essential in the perception of pain as unpleasant.4
Like other areas of the brain, the ACC has duties in addition to responding to nociceptive stimulation. One of the important roles of the ACC is in regulating attention. Another is in generating motor responses, including motor responses to pain.
The posterior cingulat cortex (PCC) is the more posterior half of the cingulate cortex. It neighbors associative and visual areas of the neocortex.
Strong stimulation of the punishment centers of the brain, especially in the periventricular zone of the hypothalamus and in the lateral hypothalamus, causes the animal to (1) develop a defense posture, (2) extend its claws, (3) lift its tail, (4) hiss, (5) spit, (6) growl, and (7) develop piloerection [hairs on end], wide-open eyes, and dilated pupils. Furthermore, even the slightest provocation causes an immediate savage attack.5
The rage phenomenon illustrates the potency of the hypothalamus in regulation of behavior. Rage is normally controlled by inhibitory signals that originate in other areas of the hypothalamus and other areas of the limbic system.
The hypothalamus is a small region of the limbic system below the thalamus that is important for homeostatic control and for connecting the brain with the endocrine system. The hypothalamus secretes hormones that in turn control the secretions of the pituitary gland, which is just beneath it. The pituitary gland secretes hormones directly into the blood stream. In this way and others the hypothalamus controls metabolic activities and other activities of the autonomic nervous system. It is involved in the generation of anti-predatory defensive behaviors.
The hypothalamus, along with other areas of the brain, continuously receives neural and chemical signals from the skin, the viscera, and deep somatic tissues. These signals are used to maintain homeostasis in the body: the core temperature, the concentration of various substances in the extracellular fluid, and the glucose concentration in extracellular fluid. Similar types of signals are used in the defense of body tissues against viral, bacterial, and other toxic challenges, including inflammation.
While it is known that there is nociceptive input to the hypothalamus (The Ascending Tracts), nociceptive input to the hypothalamus has not been well-studied.6
The insular cortex (IC on the diagram, Figure 1: Ascending paths of the pain matrix) seems to be involved in integrating sensory and motor information, emotion, and memory. Like the cingulate corter, it is adjacent to the thalamus, midbrain, and basal ganglia on its medial side. On its lateral side it is adjacent to the temporal, parietal, and occipital lobes of the neocortex. Thus the insular cortex sits between the visible neocortex and the older parts of the brain. It is part of the cortex, and is usually considered part of the limbic system.
Nociceptive activity has been consistently observed in the human IC. Electrical stimulation of the human IC has provoked extremely unpleasant painful sensations, and studies indicate that these sensations are topographically organized. This topographical stimulation of pain seems to be caused by electrical stimulation of the IC but not by stimulation to other areas of the cortex.7
The posterior portion of the IC is the part of the cortex that is most densely innervated by STT nociceptive fibers which have passed through the thalamus and are topographically organized. It is also innervated by autonomic afferents which have passed through another area of the thalamus. The IC provides an interoceptive view of the somatic and visceral state of the body. The IC then can be seen as limbic ("emotional") sensory cortex which also senses and controls the autonomic nervous system.8
Activation of the posterior IC corresponds to the strength of painful heat and cold stimulation. At least one study has shown that metabolic activation in the IC is strongly correlated with the level of pain reported by subjects.9 Lesions of the IC are sometimes associated with a condition known as pain asymbolia, in which the patient seems to sense pain normally, but behavioral and physiological reactions to the pain are abnormal. Other types of lesions may lead to central pain. (Central Pain.)
A 2009 study looked at the behavior of the brain under thermal pain in two patients who had damage to the IC on one side only. Both had higher ratings of pain intensity when the stimulus was on the side affected by IC damage than on the un-damaged side. In addition, they had higher activation of the primary somatosensory cortex (S1) under this condition, implying some reciprocal influence between IC and S1 processing.10
The anterior (front) part of the IC encodes the experienced magnitude of pain. The IC may be responsible for generating the behavior that results from painful stimulation. Studies have shown that the anterior IC is activated more strongly during acute muscle pain as opposed to cutaneous pain.11
Activation of the IC is believed to create the sensory qualities of pain and to enact homeostatic control functions that are affected by pain. At least one researcher has concluded that pain sensation is associated with a specific emotion, which is dependent on pain-evoked activity in the IC.12 (See A Rational Model of Emotion and Pain for an expansion of this idea.)
The prefrontal cortex (PFC) is part of the brain's neocortex, not part of the limbic system.
Parts of the prefrontal cortex are heavily innervated by sensory neurons through the thalamus, including nociceptive neurons. The prefrontal cortex also receives substantial input from homeostatic or bio-regulatory parts of the brain. This includes the pain-sensitive nuclei of the brain stem (The Brain Stem), the basal ganglia, the amygdala, the ACC, and the hypothalamus.13
The prefrontal cortex is also directly connected to all of the brain areas (e.g., in the hypothalamus and the brain stem) that change the state of the body, including motor output and autonomic or "chemical" output.14 Fibers from the prefrontal cortex strongly innervate the limbic regions as well as the areas of the brain stem responsible for descending pain modulation (see The Descending Tracts and Descending Pain Modulation).
Signals from external stimuli, signals from proprioception, and signals from somatic and autonomic effectors come together in the PFC. The PFC is able to connect signals associated with stimuli with signals indicating bodily state, and with the effectors of those states. Similarly, the nociceptive connections to the PFC provide a means for nociceptive signals to influence the cortex.
While limbic areas of the brain respond directly to the intensity of the pain, the prefrontal cortex responds most strongly while a sensation is just becoming painful, and it responds less strongly to higher sustained pain. In fMRI studies of chronic pain patients, it has been found that when the level of pain increases spontaneously, the brain is activated in a pattern that closely resembles its reaction to acute pain.15
A 2002 study compared the behavior of the PFC when subjected to heat-induced pain with its behavior when capsaicin was used to evoke thermal allodynia. The study showed large PFC activity in the allodynia case, along with activity in the thalamus, basal ganglia, and midbrain. Different regions of the PFC seemed to have different roles in this situation, with one area of the PFC registering the pain while another area of the PFC seemed to limit the activation of the first area.16 A separate study in 2003 found the activity in the PFC was greater in response to a painful cutaneous stimulus than to a more-unpleasant visceral stimulus.17
Thus the role of the PFC in pain generation and response is substantially different from the role of limbic and brain stem regions. The PFC has access to the entire "world" of the organism, not only its current state, but also its memories/learnings. One of its functions is to evaluate the relevance of events, including pain.18 Just as the various functional regions of the PFC can monitor and influence each other, it is believed that the PFC can monitor and limit the activity of the amygdala.19 The PFC has access to the various sensory cortices (visual, somatosensory, etc.) and it has been conjectured that it is capable of "remembering" past emotional states by controlling the state of these sensory cortices.20
(Relationships among pain, emotion, and learning are discussed in A Rational Model of Emotion and Pain.)
It is thought that the prefrontal area may be involved in activating top-down control of pain intensity. Patients who have undergone a frontal lobotomy report that they have pain, but that it doesn't bother them. (A frontal lobotomy destroys the connection from the thalamus to the prefrontal cortex. It was once a common treatment for intractable pain.) This suggests that the influence of the limbic pain areas has been diminished by the disconnection of the PFC.
The basal ganglia (Figure 3: Basal ganglia and related structures21) are a set of phylogenetically old structures that sit atop the midbrain and at the base of the forebrain. They are strongly connected to the cortex, the thalamus, the brain stem, and to other brain areas.
.svg.png)
A number of separate structures within the basal ganglia have been identified and characterized:
The caudate putamen is often found to be active in human pain studies, and the nucleus accumbens is also often active. Nociceptive neurons have been found in studies of rats and monkeys, as well as lamina V neurons.22
The basal ganglia seem to be involved in action selection. A number of behavioral disorders involve the basal ganglia, including Tourette's syndrome, Parkinson's disease, obsessive-compulsive disorder, and Huntington's disease.
Wikipedia on the basal ganglia.The amygdala is another part of the limbic system. It is part of the cortex but not part of the neocortex. It receives signals from all parts of the limbic system as well as from the temporal, parietal, and occipital lobes of the neocortex. It transmits signals back to the cortical areas from which it receives signals, to the hippocampus (which is involved in memory formation) and into the thalamus and hypothalamus.23 Although the amygdala is often referred to in common parlance as part of the "reptilian brain," this view is not phylogenetically correct. See The Triune Brain.
It is believed that the amygdala receives nociceptive input through the parabrachial area of the brain stem, although it also receives input through the thalamus. It was found that the amygdala consumed more glucose (the brain's source of energy) in studies of neuropathic pain in rats. 24
Cats that have had part of their amygdalas removed are less responsive to noxious stimuli than are normal cats. One role of the amygdala seems to be that of matching a current experience to memories of similar experiences. Monkeys that have had their amygdala removed ingest harmful objects that on the basis of their own past experience they would otherwise avoid.