Please use the form below to submit comments. Also provide an e-mail address and name. Your e-mail address and/or name will be used only to communicate with you about this or future comments you may submit. I am particularly keen to receive references to published material that contradicts the assertions and arguments I have made.
By submitting the above comment, I grant to Ross Alan Hangartner the right to incorporate the comment in full or in part, literally, paraphrased, or conceptually, as he sees fit, into State of Pain or other writings that he may create in the future. However, I don't grant permission to include my name or e-mail address, or to use them in any other way than to contact me for follow-up. I understand that by submitting the comment I acquire no right of any kind in State of Pain or other writings of Ross Alan Hangartner.
Last updated: Sat, Feb 22, 2025
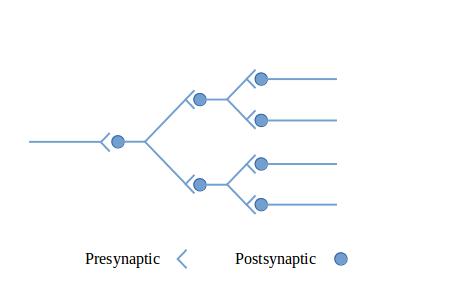
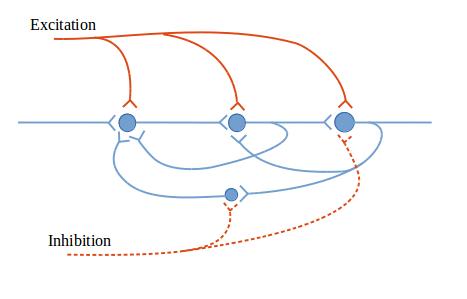
The figures in this section show circuit configurations using a simplified graphical language.1 The presynaptic terminals in these diagrams represent multiple synapses, enough to cause the effect of interest in the post-synaptic neuron. These are by no means all of the circuit types that can be made out of neurons—they are intended to give you a feeling for what neurons can do together.
Figure 1: A diverging neural circuit shows a diverging circuit. The incoming signal is multiplied by branching to more than one postsynaptic neuron. The outgoing signals could be used to multiply the strength of the signal (since a signal originally from a single neuron becomes a signal from multiple neurons), or could be routed to multiple output tracts, perhaps to distant parts of the central nervous system.

Figure 2: A converging neural circuit shows a converging circuit. The multiple inputs could come from multiple sensory neurons, or could come from other parts of the central nervous system. This circuit could fire whenever any of the inputs fired or, depending on its sensitivity, it could fire only when all three inputs fired together.
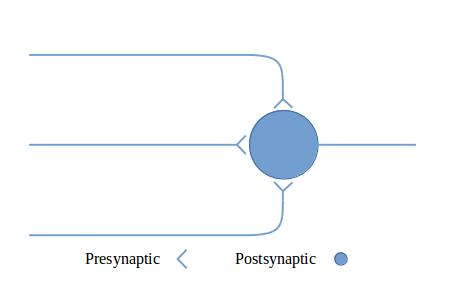
Figure 3: Exciting and inhibitory output illustrates how excitatory input can be converted into both exciting and inhibiting output by inserting a neuron with inhibitory output.
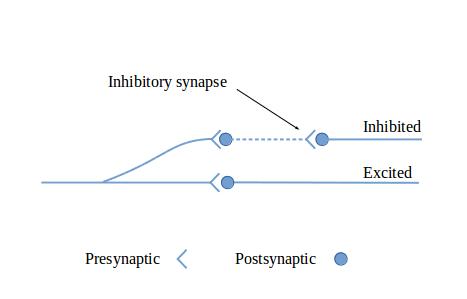
Figure 4: Reverberatory neural circuit shows a circuit that reverberates. That is, once it is excited, it can continue to produce echoes of its stimulation. This circuit includes modulating inputs. Exciting input, which is shown entering from the top of the figure, increases the output of the reverberatory neurons, while inhibiting input, shown entering from the bottom, decreases the output. Such circuits are important in nervous system functioning. A reverberating circuit can be used to produce continuous output from a brief starting stimulation; or rhythmic output, which can be modified by facilitating or inhibiting signals; or timed output.
The examples of neuronal circuit functioning in the previous section are highly simplified. Neurons are highly diverse, and their behavior is complex and can be changed by their experience. This is called neural plasticity or, as I like to say, “learning.”
Central neurons (neurons in the spinal cord and brain) have various thresholds and various response characteristics just as do peripheral or sensory neurons. They have different maximum firing rates. Some neurons are difficult to activate, while others fire continually in the absence of any external stimulation.
Synaptic strength is a term that describes how strongly a neuron reacts to a given input signal. Synaptic strength varies among neurons. It can be affected in the short and long term by excitatory or inhibitory neurotransmitters. High-frequency stimulation from presynaptic neurons can cause a long-term increase in synaptic strength, making the postsynaptic neuron more excitable. This can occur in many central neurons, including those in the spinal cord that receive their input from peripheral sensory neurons. Counteracting this is a process called presynaptic inhibition. In presynaptic inhibition, the presynaptic terminal emits neurotransmitters that affect the presynaptic terminal itself. The inhibiting neurotransmitter promotes the entry of negatively-charged ions into the presynaptic terminal endings, making the presynaptic neuron less excitable. This process is common in sensory nerve signal pathways. Adjacent sensory fibers can suppress each other, helping to prevent the spread or mixing of signals between adjacent fibers.
Fatigue is an important phenomenon in the self-regulation of neural circuits. Many neurons become fatigued when they are overused. In the short run, over-excited circuits tend to calm down because of this. In the longer term, receptors are likely to be bound if the transmitters that activate them are heavily used. Bound transmitter/receptor complexes are likely to be removed from the cell membrane by the cell. This longer-term process tends to make the neuron less excitable over time.
The repertoire of changes available to the neurons in the central nervous system includes the combination of as many as fifty different neurotransmitters with many kinds of receptors. Many sensory neurons secrete different neurotransmitters depending on their rate of firing.
Table 1 lists many of the known neurotransmitters, some of which will be familiar to you. (The names of some biochemicals reflect where they were first discovered, or what their function was believed to be when first discovered.)
| Small, quick-acting transmitters | Large, slowly-acting transmitters |
|---|---|
|
Acetylcholine Aspartate Dopamine Epinephrine (adrenalin) Gamma-aminobutyric acid (GABA) Glutamate Glycine Histamine Norepinephrine (noradrenalin) Serotonin Nitric oxide |
ACTH Alpha-melanocyte stimulating hormone Angiotensin II Beta-endorphin Bradykinin Brain-derived neurotrophic factor Calcitonin Carnosine Cholecystokinin Gastrin Glucagon Growth hormone Insulin Leucine enkephalin Luteinizing hormone Luteinizing hormone-releasing hormone Methionone enkephalin Nerve growth factor Neurotensin Oxytocin Prolactin Sleep peptides Somatastatin Substance P Thyrotropin Thyrotropin-releasing hormone Vasopressin Vasoactive intestinal peptide |
The repertoire of plasticity in spinal nociceptive circuits ranges from subtle changes in ion channel conductance to drastic changes in the morphology of cells and the connectivity and functions of neuronal networks. The various forms of plasticity include but are not limited to molecular and cellular changes in the number and functional state of synaptic, somal, and axonic ion channels, receptors, enzymes, transporter molecules, and transcription factors. This may lead to alterations in the synthesis, release, and uptake of neurotransmitters and neuromodulators; to synaptic or intrinsic plasticity; and to changes in the cytoskeleton of cells and modifications in cell morphology. Plasticity at the cellular level feeds into functional changes at the network and systemic levels, some of which ultimately lead to altered pain experiences.2