Please use the form below to submit comments. Also provide an e-mail address and name. Your e-mail address and/or name will be used only to communicate with you about this or future comments you may submit. I am particularly keen to receive references to published material that contradicts the assertions and arguments I have made.
By submitting the above comment, I grant to Ross Alan Hangartner the right to incorporate the comment in full or in part, literally, paraphrased, or conceptually, as he sees fit, into State of Pain or other writings that he may create in the future. However, I don't grant permission to include my name or e-mail address, or to use them in any other way than to contact me for follow-up. I understand that by submitting the comment I acquire no right of any kind in State of Pain or other writings of Ross Alan Hangartner.
Last updated: Sat, Mar 22, 2025
The brain contains systems that work with the spinal cord, modulating (changing or adjusting) pain perception. These are the systems that make up the gate control mechanism that was originally discovered by Wall and Melzack in the 1960s (see The Gate-control Circuits (Descending Modulation)). These systems are called “descending” because they work by sending signals down into the spinal cord. The signals can either increase (excite) the pain signals before they arrive at the brain, or can decrease (inhibit) those signals.
The balance between pain facilitation and pain inhibition is dynamic and depends upon the behavioral context. (For example, the resultant pain perception for a particular injury would be different while the organism is under threat than while the organism is resting.) It affects not only the level of pain perceived, but also the balance between nociceptive and non-nociceptive input to the brain.1 The dynamic balancing provided by this system presumably exists because the resulting behavioral variability has been biologically adaptive.

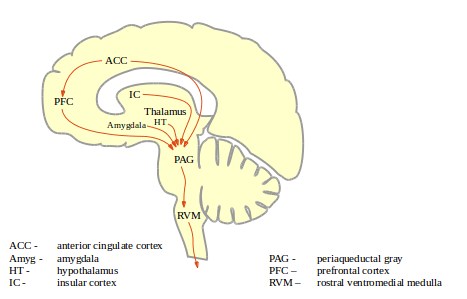
Figure 1: PAG-RVM pain pathways shows some of the brain structures that feed into the descending modulatory system. These connections allow the "cognitive, emotional, and memory" processors of the brain to influence the behavior of the pain system. The activating mechanics of the modulatory system are found mostly in two areas of the brain stem, the periaqueductal gray (PAG) and the rostral ventromedial medulla (RVM). The periaqueductal gray is an area of gray tissue (that is, heavy with neuronal bodies and dendrites) that is around (peri) a channel for cerebrspinal fluid called the “aqueduct of Sylvius.” The full name for the RVM is the description of a location in the brain stem. The PAG can be thought of as a computational center, while the RVM can be thought of as a relay center. Neurons of the RVM project down into the spinal cord where they connect with sensory neurons and with ascending projection neurons and can either excite or inhibit nociceptive sensory signals before they arrive at the brain.
It was known as long ago as the 1970s that electrical stimulation of the PAG in animals causes analgesia. This effect is specific to certain locations within the PAG. In animals, this stimulation suppressed pain behaviors (orientation, vocalizations, escape) but did not affect motor systems. Similar effects could be achieved by stimulation of the RVM. These analgesic effects were consistent across animal species, and were duplicated among humans during medical procedures. Humans reported a reduction in pain and experienced no motor effects. The preservation of this behavior across species suggests that it is of fundamental biological importance.
Subsequent research showed that PAG stimulation selectively inhibits the activity of nociceptive neurons, that is, of neurons which receive afferent input from nociceptive A-delta and C neurons. Severing the descending tracts that carry messages from the PAG interrupts these effects, and opioids recruit them.
The descending modulatory system was first believed to be a pain-inhibition system and the likely means by which opioids create analgesia. It has since been found to have both inhibitory and excitatory capabilities, and that pain facilitation is a major part of their function
2.
This system receives both "bottom-up" (spine-to-brain) and "top-down" (brain-to-spine) input. The RVM has been found to be involved in both hyperalgesia and allodynia in animal models of inflammatory and neuropathic pain, in muscle and visceral hypersensitivity, and in headache pain. Facilitation of RVM output creates a tonic aversive state
3 in rodents that manifests in increased aversion to locations in which the subject has experienced pain. These phenomena illustrate that the descending modulation system can function in a feed-forward fashion, amplifying the pain experienced in a painful condition.
The RVM is active in animal models in which mild or chronic stress cause hyperalgesia, and as part of the illness response. In these instances, the PAG-RVM system is stimulated by input from the amygdala and hypothalamus.
The system can facilitate at the same time that it inhibits pain, and it can selectively affect different parts of the body and different types of pain. It contributes to enhanced pain in conditions including inflammation, nerve injury, stress, and sickness.
This system presumably exists because it promotes the biologic fitness of the organism. One known function of the system is to enhance the sensation in an injured area while at the same time suppressing sensation from other areas, thus highlighting the painful and presumably important information of presumed damage to the self. In some circumstances (for example the presence of a predator), it might be more important to suppress painful sensation to allow the most effective reaction to the threat. Later, it may be to the benefit of the organism to emphasize the message of damage so that the organism is encouraged to rest.
In certain situations it may be adaptive to suppress reflexes that are triggered by pain, such as the withdrawal reflex (Flexor and Withdrawal Reflexes.) The RVM contains three physiologically different neuron types, which have been named ON-cells, OFF-cells, and NEUTRAL-cells. ON-cells are so-named because they turn on (fire) just before the triggering of withdrawal from noxious heat. ON-cells overall facilitate pain. OFF-cells stop firing just before a withdrawal reflex executes. OFF-cells overall inhibit pain. NEUTRAL-cells are unaffected by withdrawal. Although the human PAG-RVM system is elaborately interconnected with the brain at all levels, the behavior of these RVM cells probably demonstrates the basic behavioral purpose of the system.
Many of the RVM neurons connect not to the dorsal horn of the spine, but within the RVM. It has been found within anesthetized rats that neurons of the same type (ON-cells, OFF-cells, or NEUTRAL-cells) tend to fire at the same time
.4 So, although it is known that the descending pain modulatory system can facilitate or inhibit differently in different areas of the body, there is a tendency for them to sensitize or de-sensitize nociceptive neurons in general. When facilitated pain occurs, it increases the sensitivity of the CNS to subsequent pain, and increases the likelihood of subsequent reflex activations.
It has also been found in awake and unrestrained rats, that RVM cells similar to ON-cells respond strongly to light touch or to sudden sounds. This suggests that various stimuli that are inherently innocuous may nevertheless affect pain facilitation/inhibition. The PAG-RVM system may have a role in "tuning" perception to highlight significant incoming information. Various behavioral and autonomic variables have also been found to affect the activation of ON- and OFF-cells.5
Inflammatory substances that are released in the body following acute injury and in some chronic pain conditions can trigger an illness response which results in hyperalgesia. (See Pain and the Sickness Response.) Evidence points to the preoptic area of the hypothalamus as the likely "switch" that turns on the illness reponse. The preoptic area provides input to the PAG-RVM system, and the RVM is essential to hyperalgesia caused by the presence of endotoxins, a trigger for the illness response.6
There are several descending pain-modulating systems.7 A second system is centered on the catecholamine nuclei in the brain stem, which contain noradrenergic neurons. (The Brain Stem.) It is known that this system also is capable of both increasing and inhibiting nociception. It is not known whether this system has an effect on acute noxious pain perception. Studies on the system in chronic pain states have had inconsistent results. It is likely
that this system can be engaged separately from the PAG-RVM system, but it is also likely that the two systems interact in complex ways.8
A third descending pain-modulating system centers on the subnucleus reticularis dorsalis or SRD, an area of the medulla oblongata (see The Brain Stem). The SRD is necessary for diffuse noxious inhibitory control, or DNIC. (See Conditioned Pain Modulation (CPM) or Diffuse Noxious Inhibitory Control (DNIC)). The PAG-RVM system was initially hypothesized to be responsible for DNIC or CPM, but it was found that destruction of the PAG and key parts of the RVM did not disrupt DNIC.
SRD neurons are excited by noxious stimulation to any part of the body surface. The SRD receives ascending nociceptive input and also receives input from many parts of the brain: the PAG-RVM, the reticular formation of the brain stem, hypothalamus, amygdala, the orbital prefrontal cortex, somatosensory, motor, and insular cortex. It also sends signals to the thalamus, suggesting that it may be a channel for nociceptive input into the pain matrix.
SRD neurons descend into the outer laminae (I and IV-V) of the spine's dorsal horn. Some descending SRD neurons directly contact nociceptive neurons in the dorsal horn, which in turn project upward into the SRD. This forms a loop which functions as a feed-forward loop, magnifying pain facilitation.
Like the other descending modulatory systems, the SRD has been found to either facilitate or suppress pain, depending on how it is stimulated and depending on the organism's behavioral state.
A counter-stimulation or counterirritation phenomenon is well-documented in humans. The DNIC concept, however, of diffuse inhibitory control, has lost favor to the concept of conditioned pain modulation or CPM. Inhibition of pain by remote counter-stimulation may be the result of the PAG-RVM system rather than the SRD system.
A number of studies have shown that different CPP populations have a deficit in conditioned pain modulation, and it is possible that individuals inherently differ in the effectiveness of the CPM mechanism. Extended pain may be more likely to develop from a transient insult in those with less-effective conditioned pain modulation systems.9
The existence of multiple descending modulatory systems helps to explain the apparent contradiction between two ideas, that pain increases pain, and that pain suppresses pain. Whether the response to a given noxious stimulus is enhanced or suppressed will depend on stimulus location and duration, the environment in which the stimulus is applied, and the behavioral state of the animal.
10 Protective reflexes will sometimes be stimulated, at other times they will be suppressed.
An enormous amount of behavioral and physiological research has examined these systems, and the research permits an educated hypothesis as to what benefit the systems provide to the organism. Consider three findings based on animal models:11
1. Studies on animals have shown that inescapable noxious stimuli, predators, and situational cues associated with severe noxious stimuli can cause analgesia, which is termed "stress-induced analgesia." This same phenomenon has been demonstrated in humans. When stress-induced analgesia is conditioned by innocuous stimuli, such as a light or an aural tone, it is called "conditioned analgesia." Conditioned analgesia is triggered internally through the amygdala. These phenomena are dependent on the PAG-RVM network. They can be eliminated by lesions or by the administration of the compound naloxone, which prevents opioids from generating analgesia through the PAG-RVM system.
2. Rats can also be taught to suppress conditioned analgesia. If an innocuous signal, such as a light, is displayed at the end of a noxious conditioning stimulus, the animal learns to respond to the light as a safety signal. Showing the light in the situational context turns off the conditioned analgesia. The safety signal has a powerful and general anti-opioid effect. It interferes with the analgesic effect of systemic or injected morphine. Lesions in the RVM in this case can prevent the anti-analgesia. Injection of the neurotransmitter CCK also defeats the anti-analgesic effect of the safety signal.
3. When morphine is administered repeatedly but not continuously in a specific locational/behavioral context, it gradually loses its analgesic effect. This is called "associative opioid tolerance." Its effects are limited to the specific context in which the tolerance was developed. In other contexts, morphine retains its potency. A drug that blocks binding of a particular CCK receptor (the CCK2 receptor) blocks associative opioid tolerance. Associative opioid tolerance is associated with an increase in expression of a certain gene in an area of the amygdala.
Each of these three phenomena is an example of recognized pain-modulation processes in potentially biologically-relevant situations. (Stress-induced analgesia moderates pain while the organism is in stressful situations. Reduction of analgesia in the presence of safety signals allows the organism to return to a normal internal state. Associative opioid tolerance could be a form of homeostatic habituation.)
[P]ain modulation probably represents a more general behavioral function that implements action selection or decision making.
It is hardly unusual for animals to find themselves subject to conflicting motivations. Suppression of pain behaviors under threat can easily be imagined to be adaptive. Feeding can also be adaptive in the presence of pain. When hunger dominates, analgesia can be adaptive, and when pain dominates, hunger can be suppressed. Highly palatable food has been found to have a stronger analgesic effect in rats. This effect can be defeated by the presence of naloxone, indicating that the analgesic effect of opioids in the pain modulation systems may be responsible. Similarly, the urge to urinate competes with the urge to protect painful body parts. When a rat urinates, a subset of RVM neurons are excited, but stop firing when the urinating stops.12
The nucleus accumbens (NAc) is a region of the basal ganglia that is usually thought of as a center of reward. The NAc is connected to the hypothalamus and the amygdala, and hence to the PAG-RVM. Imaging studies on humans, however, have shown that it is activated quickly following noxious stimulation of the subject. Injection of opioids into the NAc has been found both to promote eating and suppress responses to noxious stimulation. These findings suggest that the NAc is involved in situations in which pain responses and reward-seeking motivation coexist.13
The preservation of these modulating systems across mammalian species from humans and primates through rodents to marsupials tells us that the systems have been a successful and important adaptive capability in a wide range of ecological niches. It at least suggests that the systems are capable of working effectively with or without the hypertrophied human neocortex.
Discovering the capabilities of the modulating systems has helped in understanding the great variability in pain sensation. However, much is still unknown about it, including the particular circumstances that lead to facilitation and inhibition of pain. A following section, Pain Phenomena Involving the Brain, describes phenonmena that depend upon the modulatory systems.
The psychological study of pain has historically been heavy on motivations-to-hurt and light on the adaptive motivational purposes of the pain system. This perspective on pain-as-motivation is elaborated in A Rational Model of Emotion and Pain.
Within this section...
The PAG (Last updated: Mon, Mar 3, 2025)
The RVM (Last updated: Mon, Mar 3, 2025)
Or skip to...
Neurotransmitters Involved in Pain Processing (Last updated: Tue, Mar 4, 2025)