Please use the form below to submit comments. Also provide an e-mail address and name. Your e-mail address and/or name will be used only to communicate with you about this or future comments you may submit. I am particularly keen to receive references to published material that contradicts the assertions and arguments I have made.
By submitting the above comment, I grant to Ross Alan Hangartner the right to incorporate the comment in full or in part, literally, paraphrased, or conceptually, as he sees fit, into State of Pain or other writings that he may create in the future. However, I don't grant permission to include my name or e-mail address, or to use them in any other way than to contact me for follow-up. I understand that by submitting the comment I acquire no right of any kind in State of Pain or other writings of Ross Alan Hangartner.
Last updated: Tue, Mar 4, 2025
While the spinothalamic tract (STT) largely passes through the brain stem (The Pain Matrix), the spinobulbar tracts largely terminate at various sites within the brain stem.
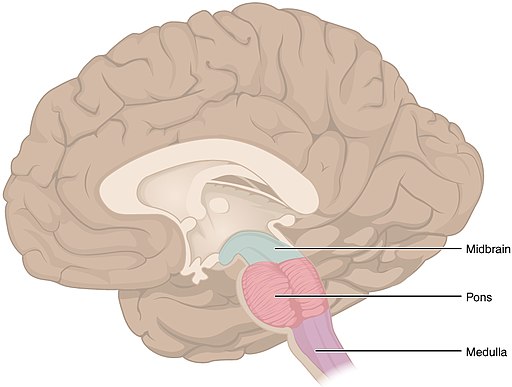
The brain stem sits atop the spine and is continuous with the spine. It is made up of the medulla oblongota, the pons, and the midbrain or mesencephalon. The forebrain in turn sits atop the brain stem. The forebrain includes the diencephalon (thalamus and hypothalamus) and the telencephalon. The telencephalon is also called the cerebrum or the cerebral hemispheres. See Figure 1: The Brain Stem1.
The brain stem is a very complex neural organ made up of a multitude of nuclei that have specialized functions, most of which operate below conscious awareness. Some of these are well understood, others not. The brain stem receives input from most of the body's sensory systems, including both somatic input (muscle, bone, and sensory organs) and visceral input (internal organs). It includes some centers that control basic homeostatic functions (temperature regulation, respiration, circulation, digestion) with limited input from the telencephalon. Other centers exert control over the body2 under control from the forebrain.

Three important functions which the brain stem performs with respect to pain are 1) regulation of the sensitivity of the spine to pain signals, 2) perception of the aching/suffering aspects of pain, and 3) regulating the level of arousal of the brain.
The regulation of spinal pain sensitivity was first demonstrated by Wall and Melzack in their gate-control theory. (See The Gate-control Circuits (Descending Modulation) and Gate Control+.) The brain stem areas most involved in adjusting spinal pain sensivity are the PAG and RVM. Their functioning is described in The Descending Tracts and Descending Pain Modulation.
The role of the brain stem in the aching/suffering aspects of pain is supported by the evidence that animals which have had their forebrain separated from their brain stem nevertheless show visible signs of suffering when they are subjected to bodily traumas. (Since the brain stem also relays motor instructions to the body, however, it is possible that such behavior is not accompanied by the perception of pain.)
The brain stem, along with areas of the hypothalamus, is involved in determining the level of arousal of the brain. Sensory signals of all kinds arouse the brain, pain signals do so particularly.
One major set of terminations for the ascending tracts is the set of catecholamine nuclei. These primarily receive lamina I terminations. The catecholamines that are most important as neurotransmitters in the brain are dopamine (DA) and norepinephrine (NE). Epinephrine, a third catecholamine, is much less important. Each of these nuclei contains thousands to tens of thousands of neurons and produces one of the catecholamine transmitters. The cells have long axons that transmit the particular catecholamine to many different areas of the brain.3
Some of the catecholamine nuclei transmit primarily within the basal ganglia. One large NE nucleus, located at the locus coeruleus and designated as A6, however, transmits to the cortex, the hypothalamus, the lower brain stem, and the spinal cord. The catecholamine cells all seem to have intrinsic pacemaker mechanisms, but are also responsive to external input. NE cells, in particular, are especially sensitive to environmental stimuli, especially those stimuli that are emotionally significant.4 (See Motive, Behavior, and Learning for the parallelism between "emotional" and "biologically significant.")
These catecholamine nuclei control the functioning of the brain holistically rather than controlling specific behavioral routines. NE seems to control sensory arousal (including autonomic), while DA seems to control motor arousal (voluntary and autonomic).
Both NE and DA seem to have a u-shaped effectiveness curve. That is, they are most effective at moderate levels of stimulation. Both overly-low and overly-high level lead to cognitive problems. Low DA levels lead to difficulty in initiating behavior. High DA levels lead to repetitive behavior. Low NE levels lead to a failure to respond to changes in the environment (sensory input is attenuated), and also lead to a tendency to act impulsively. Thus, the impulsivity of ADHD (attention deficit hyperactivity disorder) can paradoxically be limited somewhat by administration of psychostimulants.
The parabrachial nucleus (PB), an area in the pons, receives input largely from lamina 1, but with weak input from laminae IV-V. This input is only weakly somatotopic. It is highly interconnected with the reticular formation, including the catecholamine cell groups. The role of the PB is largely homeostatic and autonomic.
The PB in turn feeds into the hypothalamus, the amygdala, and several locations within the thalamus. One of the thalamic areas it touches sends signals along to the insular cortex. Nociceptive neurons that pass through the PB to the amygdala and/or hypothalamus respond to distal stimulation in the same way that lamina I nociceptive neurons do. The lamina I input through the PB thus provides a means for the brain to integrate nociceptive information from the soma with visceral, homeostatic information. It also connects indirectly to areas of the forebrain that process autonomic, neuroendocrine, and emotional information.5
The periaqueductal gray (PAG) is an area within the midbrain that surrounds one of the brains "aqueducts" that allow the circulation of cerebrospinal fluid (CSF) through the brain and spine. Spinal input to the PAG again is mostly from lamina I, but some areas also receive input from laminae VII-VIII. Some of the terminations are organized somatotopically, others are not.
The PAG is a major site for homeostatic control. When it is electrically stimulated, it can trigger aversive behaviors (such as reaction to pain), cardiovascular changes, and changes in nociceptive sensitivity, depending upon where it is stimulated and upon what behavioral state the animal is in when the stimulation occurs. The PAG works together with the rostral ventromedial nucleus (RVM) as a major actor in descending pain modulation. ([link lsect=1132). The portions of the PAG that receive spinal input also have ascending projections to both the hypothalamus and the thalamus.6
The reticular formation is an area of the midbrain characterized by a reticular or net-like organization of neurons. While nociceptive neurons have been found in areas of the reticular formation, the role of those neurons isn't known. Wall and Melzack wrote as long ago as 1988 that
The reticular formation is a particularly fascinating structure because it is superbly organized to integrate information from diverse sources and exerts a profound influence on sensory, motor and autonomic activity.7
They continued to surmise that the cells in the midbrain reticular formation would be able to sum input from spatially separate body sites, and that these cells continued to discharge following activation, so that they may carry out the function of accumulating the magnitude of pains.8 A more-recent physiology text (2006) states that
...it is likely that pain impulses entering the brain stem reticular formation, the thalamus, and other lower brain centers cause conscious perception of pain....it is believed that the cortex plays an especially important role in interpreting pain quality, even though pain perception might be principally the function of lower centers.9