Please use the form below to submit comments. Also provide an e-mail address and name. Your e-mail address and/or name will be used only to communicate with you about this or future comments you may submit. I am particularly keen to receive references to published material that contradicts the assertions and arguments I have made.
By submitting the above comment, I grant to Ross Alan Hangartner the right to incorporate the comment in full or in part, literally, paraphrased, or conceptually, as he sees fit, into State of Pain or other writings that he may create in the future. However, I don't grant permission to include my name or e-mail address, or to use them in any other way than to contact me for follow-up. I understand that by submitting the comment I acquire no right of any kind in State of Pain or other writings of Ross Alan Hangartner.
Last updated: Wed, Feb 19, 2025
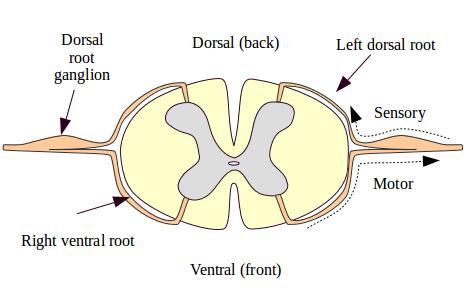
The brain receives its nociceptive input in the same general way that it receives other sensory input from the body. (But bear in mind that input isn't "nociceptive" until the brain decides that the input is painful.) Sensory neurons in the body travel through the dorsal root ganglia into the spinal cord. From there they contact spinal neurons that process the information and may transmit it up through the spinal cord and into the brain.

Figure 1: Spinal nerve roots is a cross-section of a spinal segment. It shows sensory input flowing into the dorsal horns of the spine through the dorsal root ganglia.
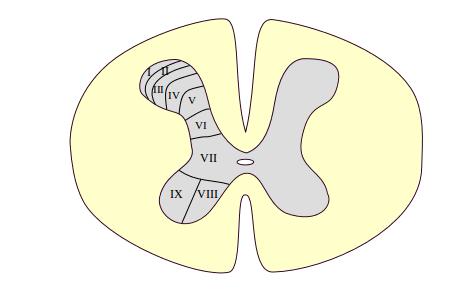
Figure 2: The laminae of Rexed shows that the spinal horns are composed of layers called the laminae of Rexed.
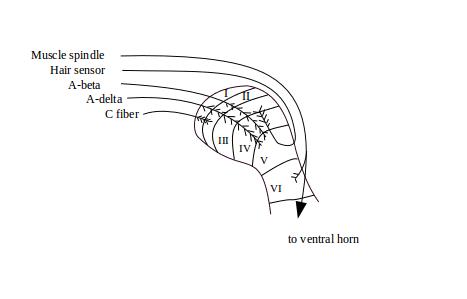
Figure 3: Arborization of afferents in the dorsal horn shows a blow-up of the right dorsal horn. The diagram illustrates that different types of neurons arborize with dorsal horn neurons at different laminae in the dorsal horn. In the laminae they are processed in various ways. (See The Spine's Organization for a review of the spine.)
Signals from the sensory neurons are fed up to the brain primarily by way of the projection neurons that originate in the gray matter of the spine. There are a number of collections or tracts of ascending fibers that follow slightly different paths and connect to different parts of the brain.
The most important of the ascending tracts for the sensation of pain, temperature, and itch are the tracts that travel up the spine, through the thalamus, and from there to various areas of the cortex. These two tracts are called the neospinothalamic tract (new) and the paleospinothalamic tract (old). The names make up for their length by being fully descriptive. Together they have an acronym, the STT.
The ascending tracts are called as "old" or "new" based on whether they also exist in mammalian species that are dissimilar to (old) or similar to (new) humans. The old tracts are slow tracts—they are made up of unmyelinated, slowly-conducting fibers that extend from the spinal segment into the brain. The new tracts are made of fast-conducting myelinated fibers. The new tracts convey much more somatotopic information than do the old. The brain can locate the source of the impulses that arrive via the new tracts much more precisely.
About half of STT cells originate in lamina I of the spinal dorsal horn, where both C and A-delta nociceptive sensory cells connect with them. About one quarter originate in laminae IV-V, and are stimulated by both A-delta and innocuous A-beta neurons. The remaining quarter originate in the ventral, motor layers of the spinal horns, laminae VII-VIII. These deep ascending neurons are large and complex cells, and are thought to inform higher neurons about the state of pools of interneurons involved in locomotion.1 There are about 10,000 STT cells on each side of the body.
The STT cells that originate in lamina I are of several types.
Taken as a group, the lamina I STT cells provide the basis for distinguishing specific types of noxious and inncocuous stimulation.2
The STT cells that originate in laminae IV-V are of other types. These primarily receive input from large-diameter, quick-transmitting A-beta neurons from the skin. Others receive input from single A-delta neurons or from multiple C neurons. Some of the lamina IV-V STT cells respond to low-threshold, innocuous stimuli such as brushing hair or graded pressure. Others respond to high-threshold signals such as those created by pinch, heat, or deep squeezing. But most STT cells from laminae IV-V are wide-dynamic-range (WDR) cells, that send smaller or larger signals up the tract depending upon how they were stimulated by the peripheral neurons.
As a group, the lamina IV-V STT cells function to integrate the input of multiple neurons and over multiple firings. Thus, they are subject (or rather, they participate in) wind-up. They are organized not somatotopically, but musculotopically, that is, by muscle. They are important in muscle reflex activity and proprioception.3
The STT cells that originate in laminae VII-VIII are again of other types. They are mostly large cells that receive input from multiple large-diameter (A-beta) neurons that originate in the skin, muscles, and joints. They are believed to generate and convey information about the relations among various parts of the body, and to be important in controlling movement.
(Certain of the cranial nerves, particularly the trigeminal nerve, serve sensory and motor functions for tissues of the face that are analagous to the functions served by the spinal nerves for the rest of the body. These cranial nerves also supply fibers into the brain that serve functions analagous to those of the various tracts that are described here.)
The terms spinobulbar and spinomedullary seem to have overlapping meanings. Both refer to spinal tracts that, like the STT, originate in the spinal segments, but ascend to one or another location in the brain stem. While the STT travels largely into the thalamus, the spinobulbar tracts travel largely into limbic areas, including the hypothalamus. These tracts are thus important for the emotional and homeostatic effects of pain.4 (This will be expanded upon in A Rational Model of Emotion and Pain.)
The spinobulbar tracts seem to originate in the same areas as the STT, that is, in laminae I, V, and VII. In addition, the spinobulbar neurons seem to have similar response characteristics to those reported above for STT neurons. This suggests that spinobulbar tract fibers have the same behaviors as STT fibers. However, as late as 2013, tracing techniques were not capable of clearly distinguishing spinobulbar from STT fibers, so it is also possible that spinobulbar neurons differ systematically from STT fibers.5 In either case, spinobulbar neurons project to active processing centers in the brain stem, as will be described in The Brain Stem.
There is some evidence of a spinohypothalamic tract or SHT. This tract originates in laminae I, V, VII, and X. Fibers ascend through the diencephalon, then descend through the hypothalamus and into the brain stem. Some of the fibers of the SHT appear to be branches of STT fibers that extend into the hypothalamus. As of 2013, no terminations of SHT fibers have been identified, but if it is in fact present in humans, this could have important implications for autonomic, neuroendocrine, and emotional effects of pain.6